The ‘gold standard’ for breast cancer detection, i.e. routine mammography, is unsustainable for low- and middle income countries, inaccessible to rural areas and requires highly trained radiologists to interpret images.
Under these circumstances, iBreastExam usefulness is emphasized here as a FDA cleared, hand-held, battery powered and fully wireless, mobile health (mHealth) solution for clinically effective breast lesion detection in low resource settings
By Sumedha Kushwaha (BDS, MDS) Head, Division of Public Health
and Garima Kumar (BDS, MPH) Product Specialist
UE LifeSciences India Pvt. Ltd.
iBreastExam for Breast Cancer Detection in Low Resource Settings
What is the global scenario of breast cancer in LMIC’s?
Breast cancer is the most commonly occurring cancer among women in India. According to the Ministry of Health, there is an occurrence rate of 25.8 per 100,000 women and mortality rate of 12.7 (Health Ministry Report). Every year around 150,000 are newly detected and 70,000 lose their life to the disease. This actually means for every 2 women newly diagnosed with breast cancer, one woman is dying of it (World Health Organization – Cancer Country Profiles, 2014). The mortality due to breast cancer is rapidly increasing because of its diagnosis at an advanced stage. As per World Health Organization (WHO), Breast Cancer accounted for 21.5% of all deaths (326,300 females) due to cancer in 2014 in India. Survival rate is poor in India with only 66.1% women surviving (2010-2014) and here more women die due to breast cancer in India than any other country. It has thus become an emergency because of its acuteness in developing countries. Projection estimates show that unless timely interventions are made, the number of breast cancer cases in India is estimated to double by 2020 (World Health Organization – Cancer Country Profiles, 2014).
Existing modalities to detect breast cancer
Self Breast Examination– It’s a highly subjective test.
Clinical Breast Examination– Subjective, a trained professional is required to carry out screening, over-burdened doctors and nurses.
Mammography– Globally, mammography is the standard technique to detect breast cancer. In India, however, because of its high cost, decreased doctor:population ratio, even reduced radiologist:population ratio, lack of trained staff, poor supply of electricity and lack of connectivity in rural areas, mammography is not a very successful technique. The machine becomes portable only when carried in a mobile medical unit, which further increases the cost. The worrisome situation is breast cancer occurrence in premenopausal women or <40 years of age, where the dense breast tissues make screening difficult, leading to the false negativity of presence of malignant lesion. The associated pain and radiation due to mammography leads to even further increased stigma attached to the disease.
Ultrasound– Requires a clinical setup with a trained radiologist. Also, there is a PC-PNDT Act in India due to high rate of female foeticide which makes the ultrasound sparsely available in rural outreach communities.
In the Indian context, there is stigma and shyness towards breast health check up. Indian females has a fear of what would be the outcome if diagnosed with cancer, acting as a major barrier to screening, early detection and treatment. Three considerable arguments that are mainly put forward by females are: personal responsibility if diagnosed with cancer, belief in and despair of the inescapability from disability and death, suspicion of cancer transmission which eventually turns out to be limiting factors in early detection.
What is the solution?
Need of the hour is to make the society believe in the ancient saying that ‘prevention is better than cure’. As breast cancer still holds the stigma, the breast health wellness test should be made acceptable to women; to accomplish this the scan should be safe, accessible and affordable. With these specifications behaviour towards health security can be changed.
The horrific statistics and the dismal response to it motivated the founders of UE LifeSciences to design a solution; here comes iBreastExam as a breakthrough to help in curbing this menace by early detection practice leading to early plus better treatment and longer survival. This is UE LifeScience’s Falgship product that finally came into existence in 2009 after years of research with a mindset that screening should not be limited to a certain population.
This is a handheld, mobile connected device, commercially available since 3 years that seeks to bring screening to areas with limited access. It was developed to quickly, easily and accurately detect abnormality in women’s breast by any healthcare giver/trained social workers. This is acceptable to women as it is painless, radiation free and present at cheaper cost as compared to other modalities thus making it a perfect pre-screening tool.
It can be kept in any primary setting as a point of care device with no requirement of any medical professional to deliver the test; making it more accessible and helping greatly in funnelling down the detected females for next line of diagnosis.
UE LifeSciences believes in providing holistic and comprehensive solution to breast cancer screening through SEWA Model. Sewa in Hindi actually means selfless service to the community. SEWA stands for Screening, Education, Wellness and Awareness led by iBreastExam.
“Every woman above age 30 should have access to routine breast health examination to ensure that breast cancer gets detected early. In India there are approximately 190 million women who can be benefitted with iBreastExam with the help of both public and private healthcare providers,” says UE LifeSciences, cofounder, Mihir Shah.
How does the health innovation product iBE work? How is it different from other screening modalities?
iBreastExam (iBE) uses patented innovative piezoelectric ceramic sensor that can assess tissue elasticity in real time. Tumours are harder than normal breast tissue and the sensors in the device can ‘feel’ them out, quickly and without any pain or radiation. UE LifeSciences trains qualified personnel employed by these healthcare providers to offer the iBE test to women who are seeking a safe and objective breast health exam. iBE provides a clinically effective, US FDA cleared, and CE marked, preliminary screening exam for breast cancer for the nearly 1 billion women around the world who have no access to a safe and reliance early screening test.
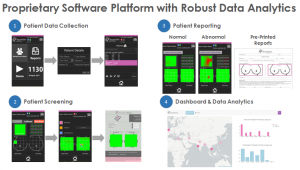
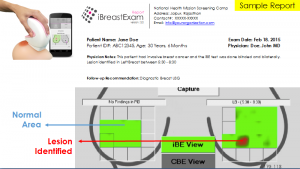
As shown in the image1, iBE software is a interface to perform breast examination, review the breast examination, store the examination data, store manual clinical examination data, document data for follow-up and print the final report. It includes data tracker also which will serve as a cancer registry: region wise, age wise and involves many more variables. This will truly develop better understanding of disease to make better health care decisions.
iBE showed sensitivity of 84% and Specificity of 94%to detect clinically relevant breast lesions. Specifically, iBE performed 19% better than an expert clinician breast exam (CBE) . As demonstrated in clinical studies iBE can objectively detect non-palpable lesion with efficacy better than CBE (Image 2).
Please find below references to the articles from global publications that have covered iBE.
- Somashekhar SP, Vijay R, Ananthasivan R, Prasanna G. Noninvasive and low-cost technique for early detection of clinically relevant breast lesions using a handheld point-of-care medical device (iBE): prospective three-arm triple-blinded comparative study. Indian J GynecolOncol. 2016;14:26.
- Broach et al. cost-effective handheld breast scanner for use in low-resource environments: a validation study .World Journal of Surgical Oncology (2016) 14:277
- Xu et al. Breast tumor detection using piezoelectric fingers: first clinical report.J Am Coll Surg. 2013 Jun;216(6):1168-73
Till now how far has the device reached?
New technologies with the potential to improve the healthcare of population through faster diagnostic and more effective care are continuously being introduced. Health care stands to benefit from the constant developments and technological innovations in life in general and in health sciences.
With recent recognition from WHO iBE has been recognised as Innovative Health Technology for low resource settings.
“With presence in 12 countries we have been able to bring breast health check-up to over 200,000 women. Our work has resulted in over 120 breast cancers being detected and those women to receive treatment early,” says UE LifeSciences, cofounder, Mihir Shah.
Regulatory Approvals
Inclusion in
WHO compendium of innovative health technologies for low-resource settings, 2016- 2017
——————————————-
About the authors
Dr. Sumedha Kushwaha (BDS, MDS, Fellow-School for Social Entrepreneurs, Fellow- Atal Incubation Centre) is a public health professional, working towards a cancer free world. She heads the division of Public Health for UE LifeSciences India Pvt. Ltd. She has authored various international research publications and books. Her spare time is usually devoted to her Not for Profit Organization called ATTAC- Aim to Terminate Tobacco and Cancer.
Dr. Garima Kumar (BDS, MPH) is currently working as Product Specialist at UE LifeSciences India Pvt. Ltd. She is passionate towards community work. She is keen to apply evidence based methods of behavioural change in community.